 
At this point, water does not have any entropy at all. If the solid water reaches absolute zero, all molecular motion ceases completely. To evaluate the enthalpy change, we can use the virial equation for the volume of a real gas, set (P0), and evaluate the resulting integral. The absolute or standard entropy of substances can. These equations relate the enthalpy and entropy of the hypothetical ideal gas standard state to the enthalpy and entropy of the real gas at pressure (P), at the same temperature. In physics, black hole thermodynamics is the area of study that seeks to reconcile the laws of thermodynamics with the existence of black hole event horizons.As the study of the statistical mechanics of black-body radiation led to the development of the theory of quantum mechanics, the effort to understand the statistical mechanics of black holes has had a deep impact upon the understanding of. As the temperature of the substance increases, its entropy increases because of an increase in molecular motion. Therefore, the entropy of a pure crystalline substance at absolute zero is defined to be equal to zero. As the water is cooled further, bringing the temperature closer to absolute zero, the vibration of the molecules diminishes. All molecular motion ceases at absolute zero (0 K) ( 0 K). The ice molecules can no longer move freely but can only vibrate within the lattice. Water cools further, resulting in solid ice. 
During the condensation process, they lose some entropy. The molecules can still move around, but not as freely as gas. Water in its gaseous state has molecules that can move around very freely – it has high entropy. One of the most intriguing equations of contemporary physics is Boltzmann’s celebrated principle S B k B ln W, 1 where k B is Boltzmann’s constant. 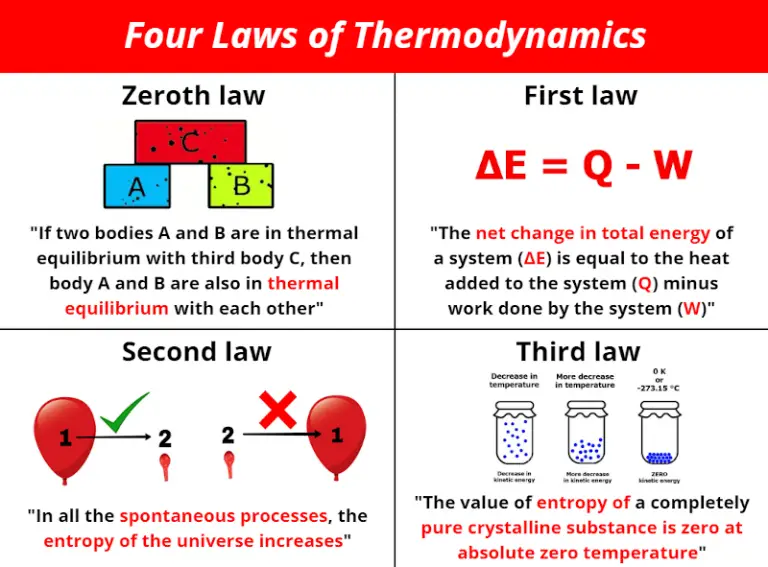
We can take the example of water to demonstrate the third law of thermodynamics. Thus, a perfect crystal’s entropy at 0 K is zero. According to Boltzmann equation, the entropy as a function of the number of microstates corresponding to each macrostate (W) isĪ perfect crystal has only one unique ground state. It means the system has only one accessible microstate, the ground state. All atoms and molecules are at their lowest energy points. Mathematically, it is written as follows :Īt zero Kelvin, the system does not contain any heat. EquationĪccording to the third law of thermodynamics, the entropy change for a physical or chemical transformation approaches zero as the temperature approaches absolute zero. 2Freeexpansion Anexamplethathelpselucidatethedi erentde nitionsofentropyisthefreeexpansionofagas fromavolumeV 1toavolumeV 2. For impure crystals, or those with faulty alignment, some energy will be associated with the imperfections, so the entropy cannot become zero. According to Nernst, “it is impossible for a process to bring a given system’s entropy to zero in a discrete and finite number of steps.” Third Law and Perfect CrystalĪ perfect crystal is one in which every molecule is undistinguishable, and the molecular alignment is flawless throughout the substance. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
